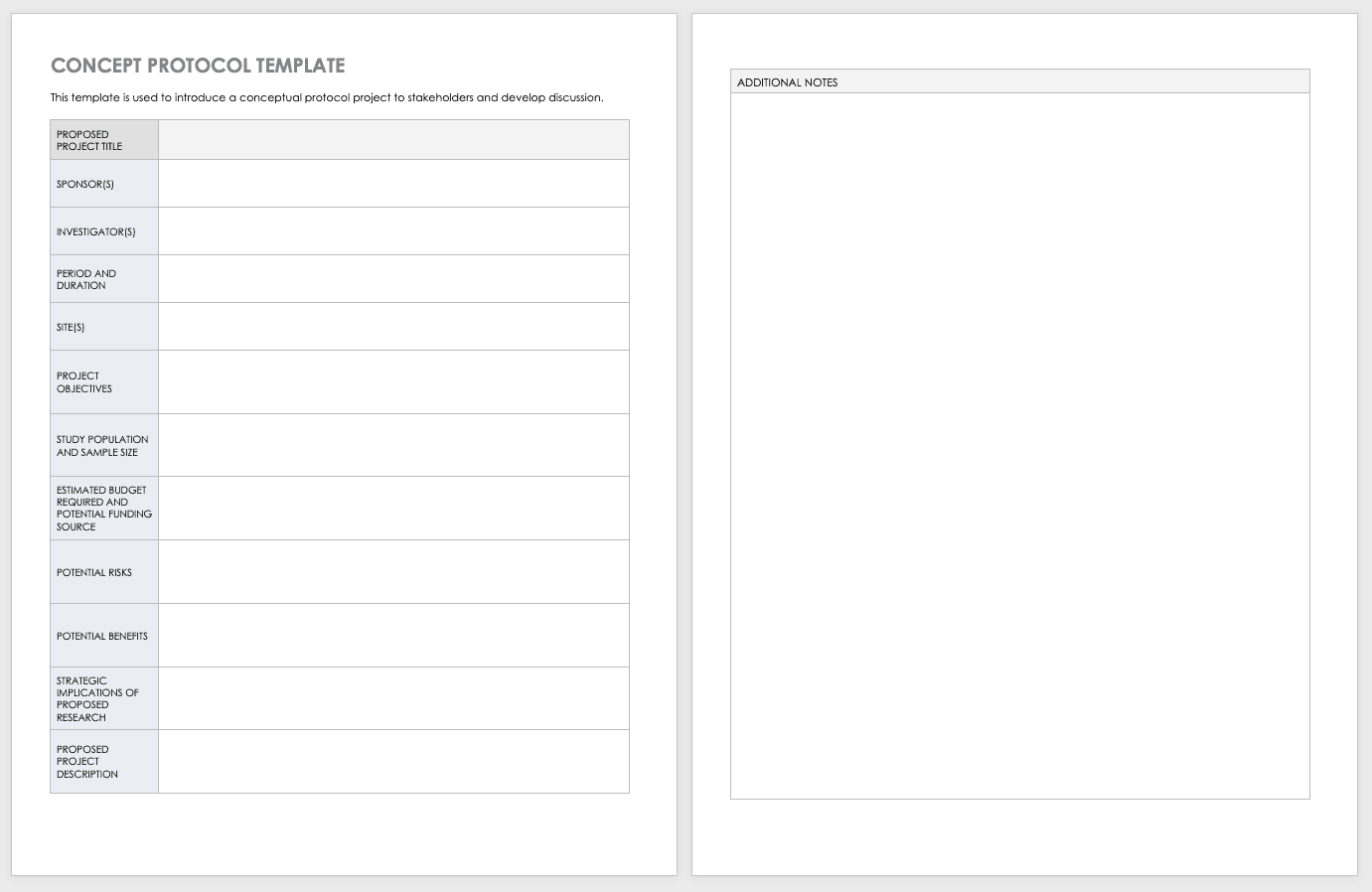
NIH-FDA Phase 2 and 3 IND/IDE Clinical Trial Protocol Template PREFACE Remove this Preface before finalizing and distributing

White Paper: Protocol Design in Real-World Evidence: The Indispensable Link Between Strategic Need and Study Execution - Evidera
TEMPLATE FOR THE CLINICAL ASSESSMENT OF CLINICAL TRIAL APPLICATIONS African Vaccine Regulatory Forum (AVAREF) CLINICAL ASSESSMEN
Research Study Protocol Template Instructions Study Protocol Title: Table of Contents: List of Abbreviations: Principal Investig