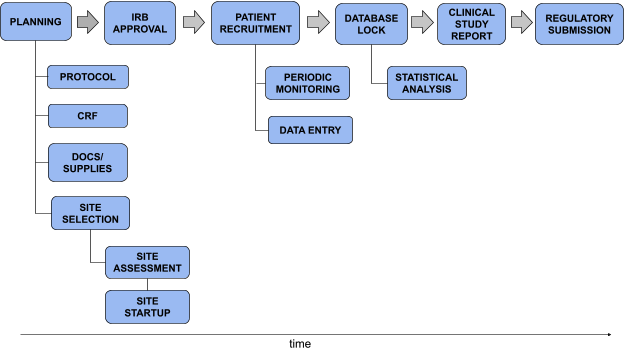
Services - Clinical Trial Report Preparation Services from Lucknow Uttar Pradesh India | ID - 3118976

Example of Clinical Trial Assessment of Infrastructure Matrix (CT AIM)... | Download Scientific Diagram

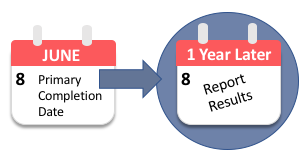
Compliance with legal requirement to report clinical trial results on ClinicalTrials.gov: a cohort study - The Lancet
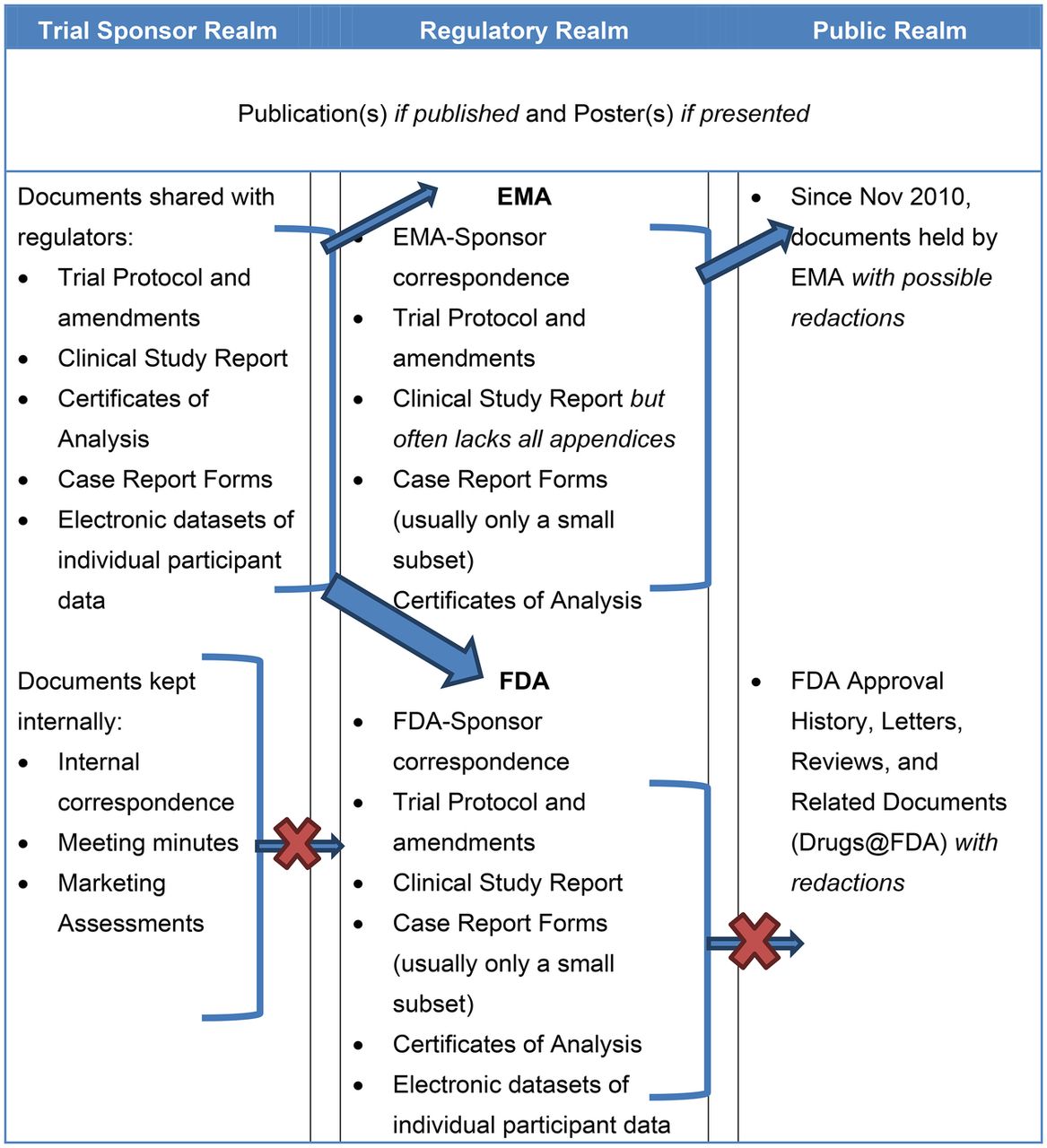
Clinical study reports of randomised controlled trials: an exploratory review of previously confidential industry reports | BMJ Open

Construction of study sample comprising the 50 most recent clinical... | Download Scientific Diagram
Completeness of Reporting of Patient-Relevant Clinical Trial Outcomes: Comparison of Unpublished Clinical Study Reports with Publicly Available Data | PLOS Medicine
Completeness of Reporting of Patient-Relevant Clinical Trial Outcomes: Comparison of Unpublished Clinical Study Reports with Publicly Available Data | PLOS Medicine

Will clinical trial data disclosure reduce incentives to develop new uses of drugs? | Nature Biotechnology

Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension - The Lancet Digital Health

Clinical Trial Report Template (6) - TEMPLATES EXAMPLE | TEMPLATES EXAMPLE | Report template, Clinical trials, Template google

Appendix F Illustrative Data Fields for the Results Summary | Developing a National Registry of Pharmacologic and Biologic Clinical Trials: Workshop Report | The National Academies Press

![PDF] Effective authoring of clinical study reports: A companion guide | Semantic Scholar PDF] Effective authoring of clinical study reports: A companion guide | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/2f8d38c9afbd7fce7a7635cf459a7989c82a753a/5-Table1-1.png)