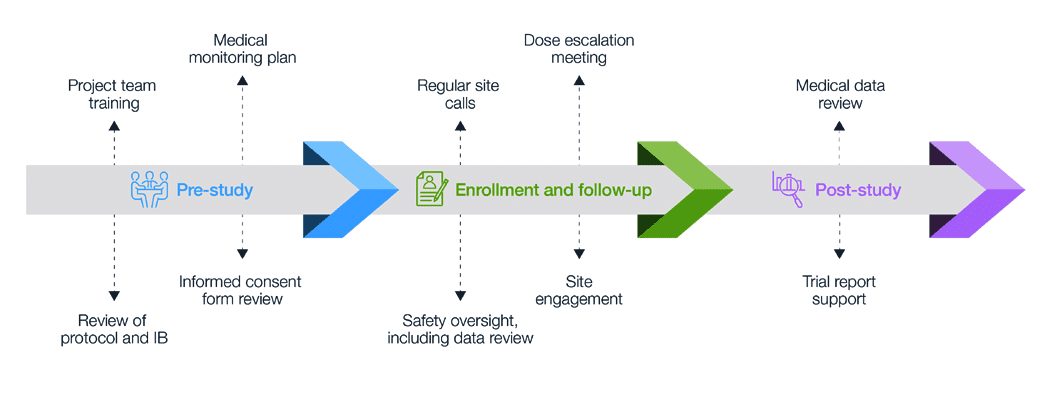
Data Safety and Monitoring Boards Should Be Required for Both Early- and Late-Phase Clinical Trials - ScienceDirect
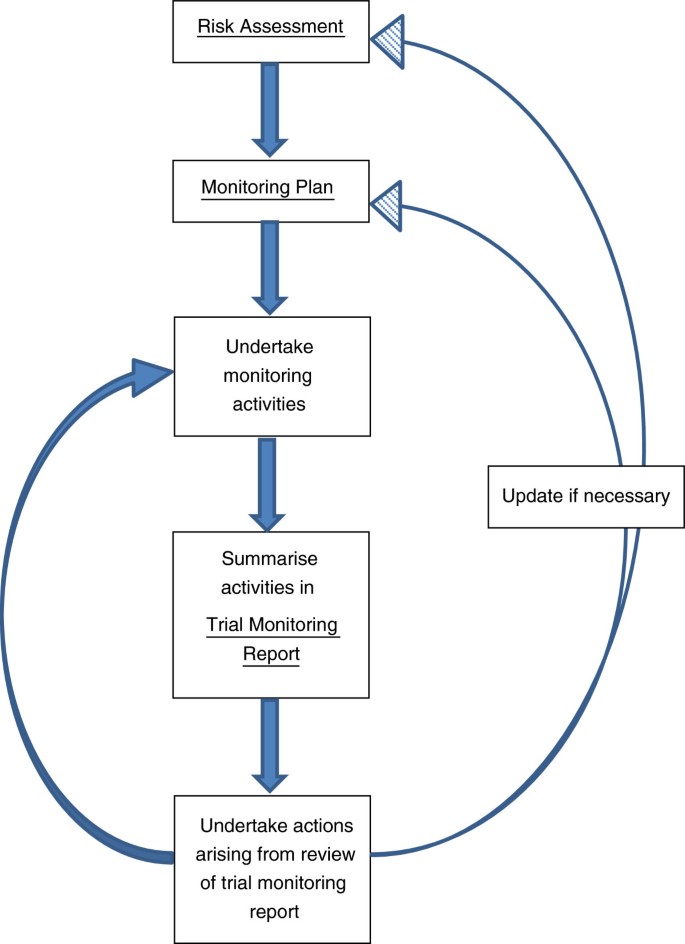
Risk-proportionate clinical trial monitoring: an example approach from a non-commercial trials unit | Trials | Full Text

Evaluation of safety and immunogenicity of receptor-binding domain-based COVID-19 vaccine (Corbevax) to select the optimum formulation in open-label, multicentre, and randomised phase-1/2 and phase-2 clinical trials - eBioMedicine

The flowchart of the Two-stage Bayesian safety monitoring for blinded... | Download Scientific Diagram
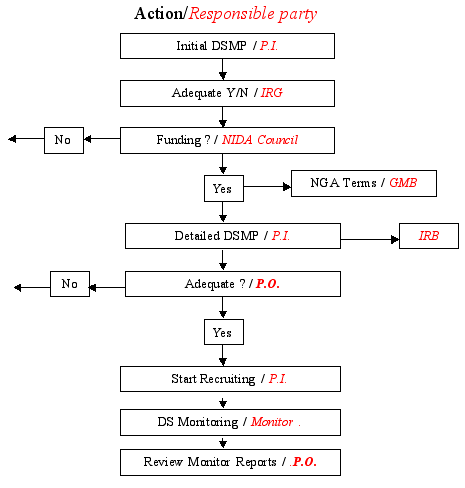
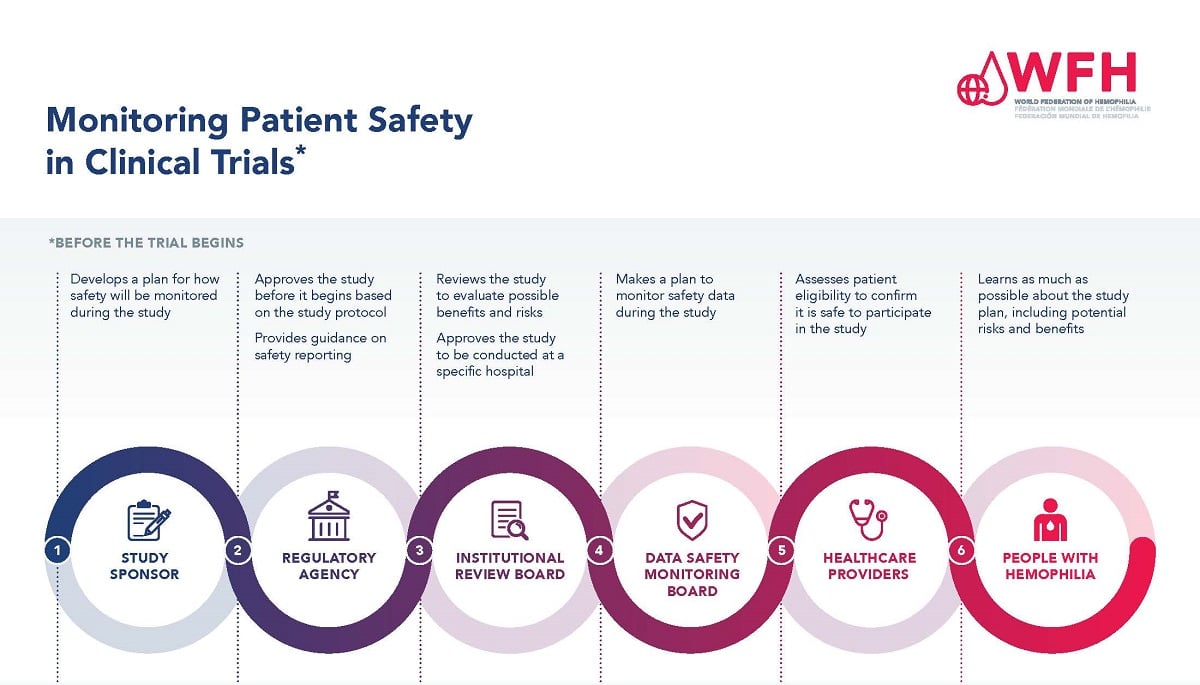
Guidelines for Developing a Data and Safety Monitoring Plan | National Institute on Drug Abuse (NIDA)

Safety Monitoring and Adverse Event Reporting in Clinical Trials: Regulatory Requirements and Best Practices

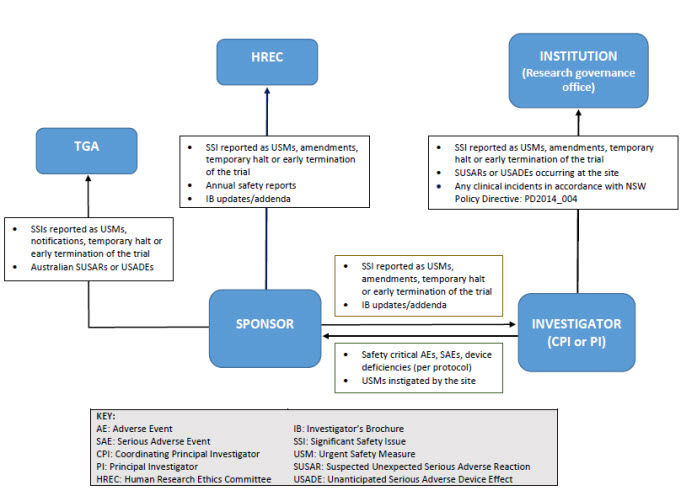








![PDF] Data and Safety Monitoring Board and the Ratio Decidendi of the Trial | Semantic Scholar PDF] Data and Safety Monitoring Board and the Ratio Decidendi of the Trial | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/95d78007a60d58184bdcea808f1f26f279ce7078/23-Table1-1.png)